 Thus, water is H2O and carbon dioxide is CO2. By convention, no subscript is written when a molecule contains only one atom of an element. In most cases, they have a relatively simple crystal structure distinguished by a close packing of atoms and a high degree of symmetry. In the following example, the Book element definition includes the Chapter element. Britannica Quiz Building Blocks of Everyday Objects liquid metal See all videos for this article Metals are usually crystalline solids. The shorthand notation for a compound describes the number of atoms of each element, which is indicated by a subscript written after the symbol for the element. Sub-elements of these inserted definitions can also be defined locally. Nonmetals combine with each other to form covalent compounds, which exist as neutral molecules. a small amount of an emotion or quality: There was certainly an element of truth in what she said. These compounds are composed of positive and negative ions formed by adding or subtracting electrons from neutral atoms and molecules. Metals often react with nonmetals to form ionic compounds. When a compound decomposes, the atoms are recovered unchanged Compounds Elements combine to form chemical compounds that are often divided into two categories. Atoms of different elements combine in simple whole numbers to form compounds. Atoms of different elements have different weights and different chemical properties. Matter is made up of atoms that are indivisible and indestructible. Atoms Elements are made up of atoms, the smallest particle that has any of the properties of the element.John Dalton, in 1803, proposed a modern theory of the atom based on the following assumptions. The semimetals can be found along the dividing line between the metals and the nonmetals. A handful of nonmetals are clustered in the upper right corner of the periodic table. Most elements are metals, which are found on the left and toward the bottom of the periodic table. The elements can be divided into three categories that have characteristic properties: metals, nonmetals, and semimetals. 1: Language The most important literary element is language. The notation for each element can be found on the periodic table of elements. Each term in the literary elements list below gives you the literary element definition and an example of how the elements work. Each element is represented by a unique symbol. They are therefore the elementary, or simplest, chemical substances - elements. Hydrogen and oxygen, on the other hand, cannot be decomposed into simpler substances. Discover the element definition in chemistry, what makes an element. Example: Water decomposes into a mixture of hydrogen and oxygen when an electric current is passed through the liquid. Transcript Author Nicholas Amendolare View bio Instructor Marion Carroll View bio Learn about elements in chemistry. Because atoms cannot be created or destroyed in a chemical reaction, elements such as phosphorus (P4) or sulfur (S8) cannot be broken down into simpler substances by these reactions. A program could also use an external source such as a file or a database to create a list of elements.Any substance that contains only one kind of an atom is known as an element. This example is one way elements could be added and used in a program. In the example above, the following output would be printed to the screen. Foreach array example my = ('Neil', 'Geoff', 'Nathan', 'Scott', 'Tim') 
For example, in the below Perl code the array contains five elements (names) and then uses a foreach to say hello to each of the names. For example, in computer programming an array can contain different elements ( index) that can be stored and called upon individually. An element is a single part of a larger group. 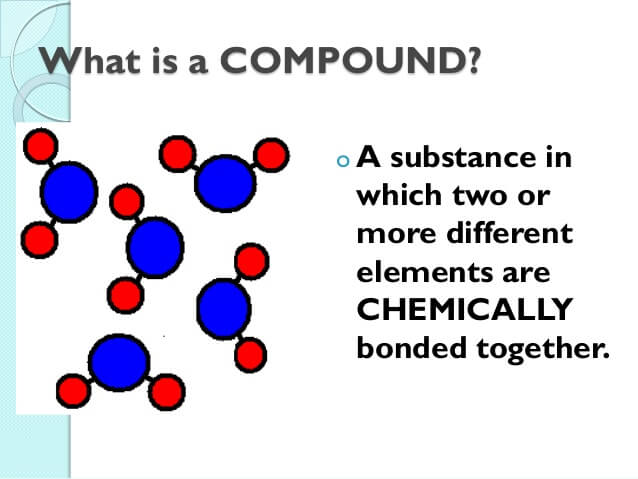
Elements is used as an abbreviation of Adobe Photoshop Elements.ģ. When referring to Hypertext Markup Language, see our HTML element definition for further information.Ģ. 
An element may refer to any of the following:ġ. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
